Quick Details
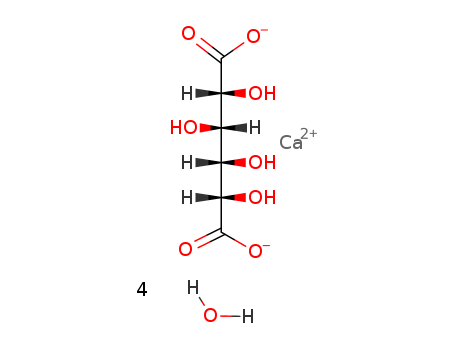
- ProName: 4-O-beta-D-Galactopyranosyl-D-gluconic...
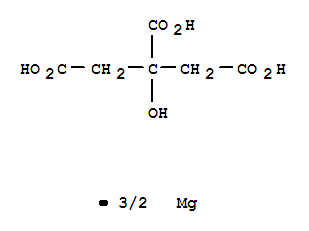
- CasNo: 110638-68-1
- Molecular Formula: 2(C12H21O12).Ca....
- LimitNum: 0
Superiority
Analytical Specifications for Calcium Lactobionate USP (Injection grade - in house specs.)
Tests Specifications:
Description : White to off- w…
Details
Analytical Specifications for Calcium Lactobionate USP (Injection grade - in house specs.)
Tests Specifications:
Description : White to off- white ,finely crystalline powder, may be partially granular, Freely soluble in water, odorless.
Solubility
: Soluble in water. Freely soluble in boiling water
Identification
a) Calcium
b) IR Spectrum
c) TLC
Shall pass the test
Shall pass the test
Shall pass the test
Clarity of 20% w/v solution in boiling water : Clear to nearly clear
Colour of 20% w/v solution in boiling water : Colourless to nearly colourless
Behaviour on sterilization 20% w/v solution : Clear, colourless, not more coloured than the colour comparison solution.
pH of the solution (1 in 20) : Between 5.9 and 7.5
Reducing substances : Max. 1.0%
Specific rotation : Between + 22.00 and + 26.50
Chloride
: Max. 50 ppm
Sulphate : Max. 50 ppm
Heavy Metals
: Max. 5 ppm
Iron : Max. 10 ppm
Phosphate : Max. 100 ppm
Arsenic Max. 1 ppm
Alkali metals and magnesium : Max. 0.2%
Organic Volatile Impurities
a. Ethanol
b. Acetone
:
Max.1000 ppm
Max.500 ppm
Oxalic acid
: Max.100 ppm
Assay : 98.0% -102.0%
Calcium content : 4.97 % - 5.17%
Particle Size Distribution
Passing thru’ U.S 4#
:
More than 90 % w/w
Bulk density (Loose) : 200 - 500 kg/ m3
Microbial Limit Tests:
Bacterial Endotoxin : Max. 0.03 EU (IU)/mg
Total viable aerobic count : Max. 100 CFU/g
Total fungus
: Max.50 cfu / g
Enterobacteriaceae
: Absent in 1g
E.Coli : Absent in 10 g
Salmonella : Absent in 10 g
Staphylococcus aureus : Absent in 10 g
Pseudomonas aeruginosa : Absent in 10 g
1. Method of testing as per USP 29
2. In house specs - stringent limits than USP 29 + additional in house specs. - Method of testing available on request.
3. MSDS - Available on request.
Other:
Shelf life : 3 years from date of manufacturing.
Storage condition : Preserve in a sealed container at temperature between 20-250C.
Manufacturing Condition : WHO cGMP approved.
Quality Certification : ISO, USFDA, EDQM, cGMP, EU-Compliance, HACCP,
KOSHER – StarK PAREVE Status, HALAL & ICH-Q7A.
Packaging Units : Products can be supplied in packaging ranging from
20 kgs – 50 kgs
Packing : Packing Material options include HDPE Bags Plastic Drums,
Fiber Drums, Fiber Kegs, High Resistance Paper Sacks and High Density Carton with inner HDPE liner sealed air tight.
Dated : 25th September 2006