Quick Details
- ProName: Calcium gluconate
- CasNo: 299-28-5
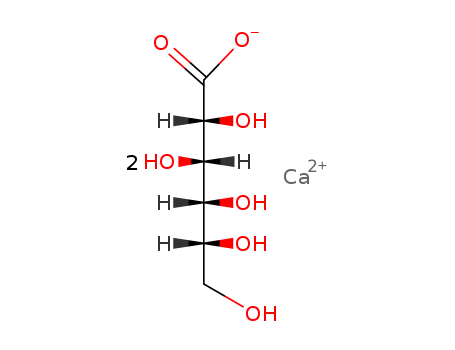
- Molecular Formula: 2(C6H11O7).Ca
- LimitNum: 0
Superiority
Analytical Specifications for Calcium Gluconate USP (Injectable grade) (Anhydrous/ Monohydrate)
Tests Specifications:
Characters : A white crystalline or granular powder.
Solubility : Sparing…
Details
Analytical Specifications for Calcium Gluconate USP (Injectable grade) (Anhydrous/ Monohydrate)
Tests Specifications:
Characters : A white crystalline or granular powder.
Solubility : Sparingly (slowly) soluble in water, freely soluble in boiling water
Identification
TLC
Calcium
:
: Shall pass the test.
Shall pass the test.
Loss on drying
: Max. 3.0 % (Anhydrous)
Max. 1.0 % (Monohydrate)
Chloride
: Max.0.005%
Oxalate : Max.0.01%
Phosphate : Max.0.01%
Sulfate : Max.0.005%
Arsenic : Max.3 ppm
Heavy metals : Max.0.001%
Magnesium and alkali metals : Max.0.4%
Iron : Max.5 ppm
Reducing substances : Max.1.0%
Organic volatile impurities : Shall meet the requirements
Residual solvents : Shall meet the requirements
Assay as C12 H22 CaO14 (on dried basis)
: 98 .0 % -102.0% (Anhydrous)
Assay as C12 H22 CaO14H2O : 99.0 % - 101.0% (Monohydrate)
Additional tests (In-house)
Clarity of 10% w/v solution in boiling water Clear
Colour of 10% w/v solution in boiling water Colourless
Calcium content (on as is basis) 8.85 % - 9.50% (Anhydrous)
8.85 % - 9.03 % (Monohydrate)
pH (5% solution) App.7.0
Lead Max.0.2 ppm
Mercury Max. 1 ppm
Particle Size Distribution
Passing thru’ U.S 16 # More than 70 % w/w
Bulk density (Loose) 250 - 600 kg/ m3
Microbial Limit Tests (in- house specs.)
Bacterial Endotoxin Max.0.075 EU / mg
Total viable aerobic count Max.100 cfu /g
Total fungus Max.50 cfu / g
Enterobacteriaceae Absent in 1 g
E.Coli Absent in 10 g
Salmonella Absent in 50 g
Staphylococcus aureus Absent in 10 g
Pseudomonas aeruginosa Absent in 10 g
1. Method of testing as per USP 29
2. In house specs. - Method of testing available on request.
3. MSDS - Available on request.
Other:
Shelf life : 3 years from date of manufacturing.
Storage condition : Preserve in a sealed container at temperature between 20-250C.
Manufacturing Condition : WHO cGMP approved.
Quality Certification : ISO, USFDA, EDQM, cGMP, EU-Compliance, HACCP,
KOSHER – StarK PAREVE Status, HALAL & ICH-Q7A.
Packaging Units : Products can be supplied in packaging ranging from
20 kgs – 50 kgs
Packing : Packing Material options include HDPE Bags Plastic Drums,
Fiber Drums, Fiber Kegs, High Resistance Paper Sacks and High Density Carton with inner HDPE liner sealed air tight.
Dated : 25th September 2006